Servo Magazine ( April 2012 )
Robot-Assisted Prostate Surgery
By Tom Carroll View In Digital Edition
Cancer! That’s a word we all dread to hear, whether about someone we know or especially concerning ourselves. I heard that word spoken to me early last year after I had a prostate biopsy. Needless to say, thousands of things were spinning through my mind. I knew that prostate cancer is the second most common cancer among men worldwide, with one in six men here in the US diagnosed with the disease. Another scary fact is that prostate cancer is the second leading cause of cancer deaths among US men. The good news is if the cancer is discovered early, there is a much greater chance of curing prostate cancer. “What is the next step?” I asked my doctor, as I was aware that there were many options available to men with this condition.
As a reader of a robotics magazine, I’d like to take you on a trip into an operating room with a robot performing surgery on a person: me. Before discussing robot-assisted prostate surgery, I’d like to go back a bit to my first prostate biopsy back in 2002. Understanding prostate health is important in understanding the need for any sort of treatment option. I had a PSA (prostate specific antigen) blood test score of 4.7 and it had been slowly rising from 3.2 to 3.4 in the late 1990s. My family practice doctor was concerned and sent me to a urologist. With data in hand, my urologist performed the needle biopsies and he found no cancer. Yes, the days waiting after the biopsy until he told me the ‘good’ results were not days that I enjoyed. Nine years later and a new doctor, my PSA had risen to 11.95 and another needle biopsy was necessary.
There has been much speculation about the need for a PSA test recently, and even some doctors raised concern that the test could actually be harmful since a few patients have incurred severe infections from the biopsy procedure. Some patients have been rushed into surgery only to find tiny scattered cells that would not have grown into tumors for years. I happen to be in the majority who believe in these tests and the subsequent biopsies. The needle biopsies are not enjoyable but can discover a cancer situation where subsequent surgical procedures can be performed to save a life — my life.
The Options
There are many options that urologists have at their disposal, though most of these specialists prefer to use only one or two of them in the treatment of their patients. One, of course, is to wait and see. Prostate cancer is usually a very slow growing type of cancer and many times a man may die of some other age-related disease before the prostate cancer could ever be fatal.
Standard surgical removal of the prostate and seminal vesicles via the tried and true open surgery method is also an option, though the hospital recovery period is longer. Doctors can also use laparoscopic methods wherein smaller incisions are used to enter the body at the site of the prostate, and handheld small instruments are then inserted, along with a light and vision system (fiber optic or television) to visualize, manipulate, and cut and remove tissue. Robotic surgery is similar to laparoscopic, but better control and magnification is possible.
Radiation therapy — either implanted radioactive seeds or external beam — to kill the cancerous tissue is another method. Cryotherapy is used since it does kill the complete prostate gland and any cancer cells, but it has some severe side effects. Hormone therapy and chemotherapy are used in many cases; a good friend of mine swears by his treatments.
Your doctor will discuss these options with you and may strongly suggest the method he/she prefers. My doctor chose to completely remove my cancerous prostate and the nearby seminal vesicles as my treatment option. If you find yourself facing any sort of cancer possibility and feel uncomfortable with the methods your doctor discusses, of course, get a second opinion.
Robot-Assisted Prostate Surgery
Six years ago, I wrote an article for this column about robotic surgery, having suspected a possibility of having prostate cancer sometime in the future after my first biopsy in 2002. I began looking at various options and quickly settled on a robot-assisted prostatectomy as the best choice for me. The choice was not made because of my robotics background, but due to the less invasive method employed. I found out that my present urologist (Dr. Michael Lipke) had previously trained on the Intuitive Surgical da Vinci system shown in Figure 1 and had performed hundreds of successful prostate surgeries.
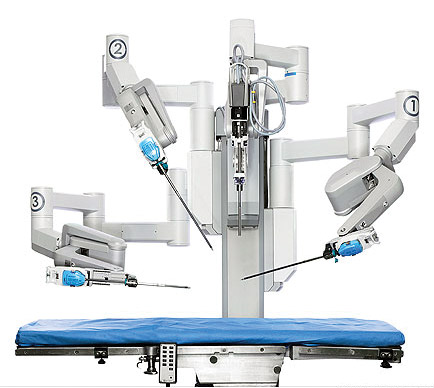
FIGURE 1. Intuitive Surgical da Vinci robot.
Despite the terminology, the da Vinci ‘robot’ is not a true intelligent or autonomous robot, but is a tele-robot controlled by a surgeon from a nearby console (see Figure 2); as the title implies, it’s robot assisted surgery.
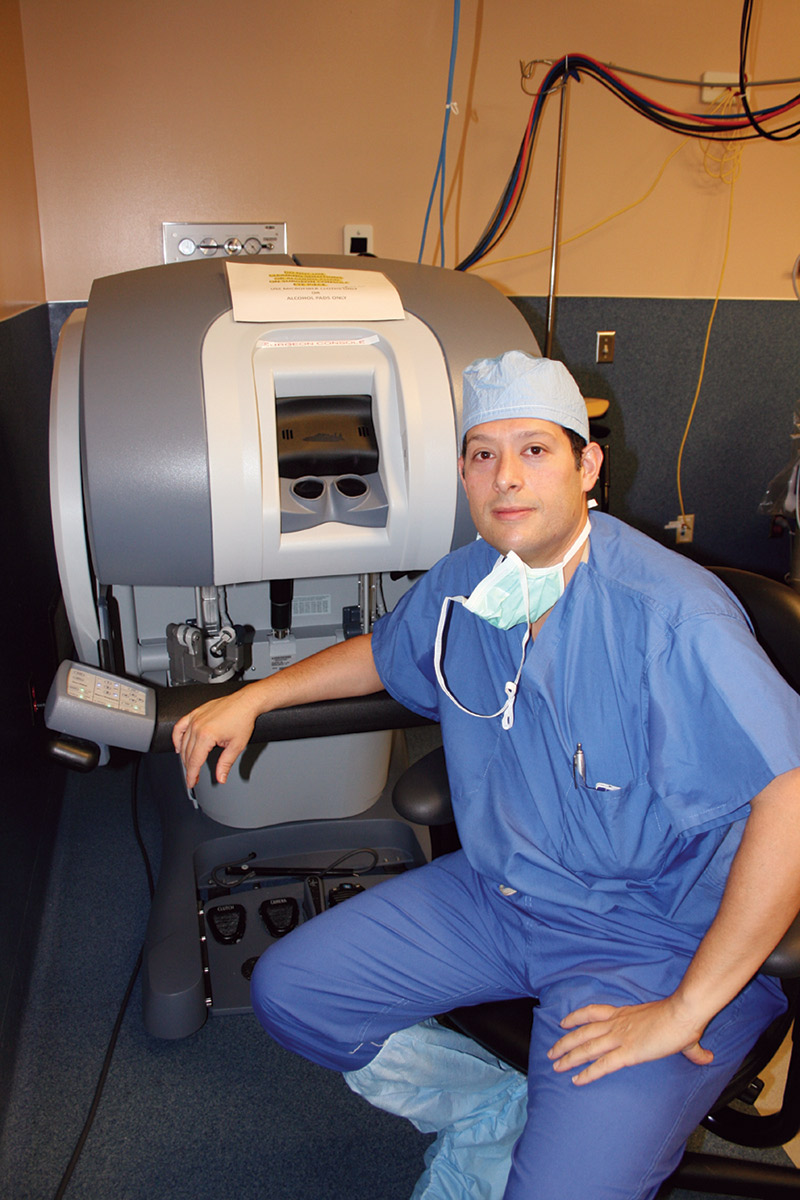
FIGURE 2. Michael Lipke, MD at the console of the da Vinci system.
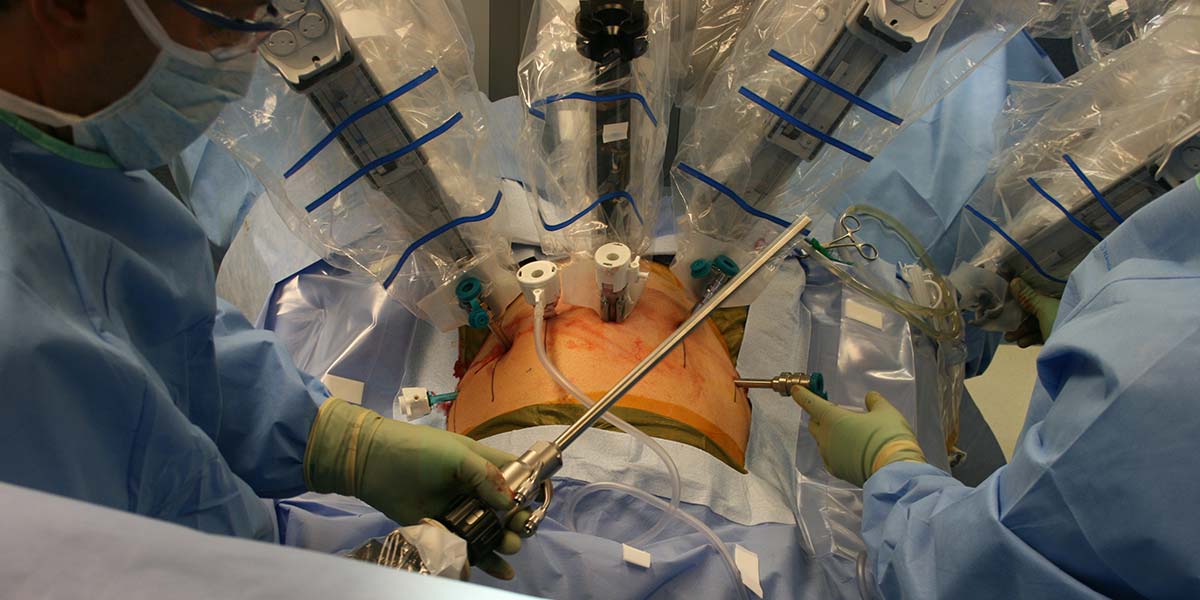
Figure 3 shows the general operating room setup. My surgeon used the da Vinci “S” HD system, though the hospital also had a newer da Vinci “Si” surgical system.
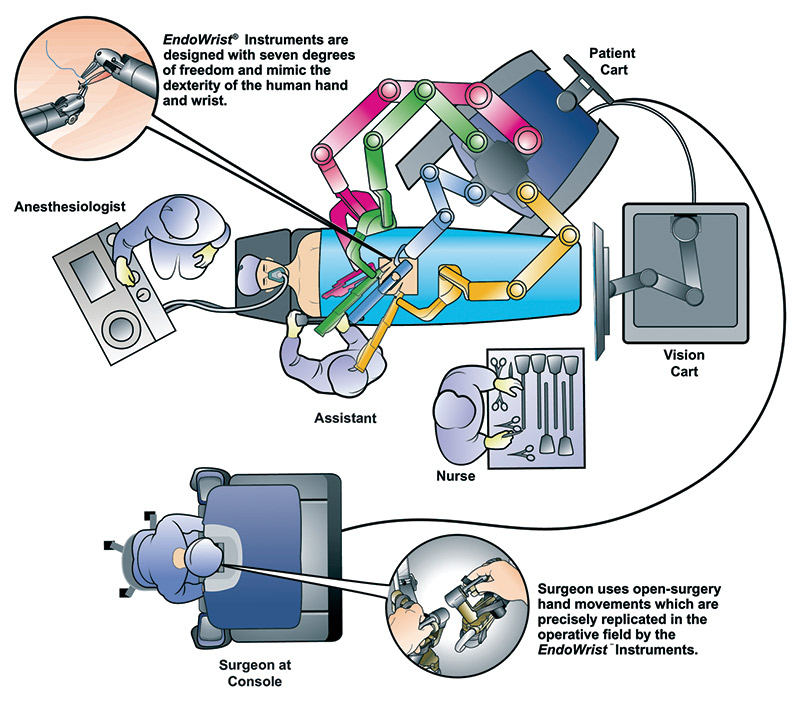
FIGURE 3. da Vinci surgical system operating room setup.
Training to use the da Vinci system is getting easier. Mimic Technologies is developing MScore — a portable trainer device shown in Figure 4.

FIGURE 4. Mimic Technologies da Vinci trainer.
As stated from MScore: “The MScore system allows every movement and action the <prospective> surgeon makes to be tracked and evaluated within a virtual reality training environment. A surgeon’s proficiency and score is established by utilizing a wide variety of performance metrics, such as task time, efficiency of instrument motion, blood loss, and the force applied to tissue. Performance baselines are derived from the data collected from experienced surgeons.” This type of trainer allows surgeons to practice and improve without taking up valuable and expensive time on the actual machine.
How are Various Surgeries Performed?
To understand robot-assisted surgery, you must sit back and imagine how surgeons perform any type of surgery on a human. In typical open procedures, the area to be operated on is fairly wide open and exposed, with clamps to hold tissue in place and to make it easier to apply suction to remove fluid buildup. A large overhead lamp illuminates the area. The surgeon can then use scalpels, clamps, forceps, hemostats, and other instruments that can easily be manually manipulated in whatever way necessary. The surgeon is usually standing up and looking down on the surgery site.
Now, imagine a more localized surgery such as a shoulder or knee joint where the surgeon elects to use arthroscopic techniques to operate through small holes in the skin and tissue. Arthro meaning ‘joint’ and scopic meaning ‘vision’ surgery is a technique whereby the doctor uses handheld instruments to operate within the body without performing major open surgery; a few of which are shown in Figure 5.

FIGURE 5. Ankle arthroscopy instruments.
Figure 6 shows knee surgery using a cannula to circulate saline solution within the operating area, an arthroscope to allow the surgeon to see within the joint area, and the particular surgical instrument.
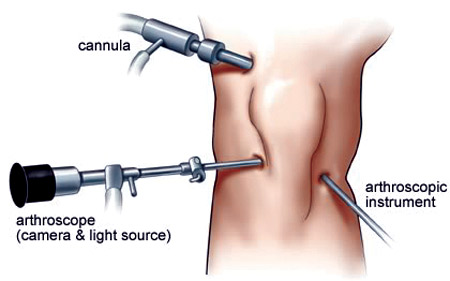
FIGURE 6. Arthroscopic knee surgery.
Sometimes the term orthoscopic surgery is used, but this is a broad term implying clear vision surgery. As you can imagine, the surgeon can maneuver the instrument around enough to get to the small areas required for surgery. The outer skin and tissue can move a bit to allow entry from slightly different directions. This type of surgery is also called laparoscopic surgery (or minimally invasive surgery; MIS) when it is performed in other areas of the body.
The Case for Robot-Assisted Surgery
Now, envision an operating site deep with the body — a location such as the prostate and bladder — that could only be reached by open surgery techniques. The depth of the surgery prevents ease of movement with the arthroscopic instruments without making large skin/tissue incisions. The surgeon now requires many axes of instrument movement — not available with handheld arthroscopic instruments. If a particular surgery requires 90 degree positioning of scissors or forceps to reach a particular area of interest, the surgeon could enter from another hole in the body, or use an instrument that can rotate at the end with several degrees of freedom (such as many industrial and experimental robot arms). This is the technique practiced by surgeons using the da Vinci system.
With the operating site so deep within the patient’s body and only small holes in the skin and underlying tissue available, the surgeon is limited in how much he or she can actually move the ends of the EndoWrist instruments — the working ‘hands’ of the robotic system (more on this in a moment).
Many months of intensive training is required before a surgeon begins to feel at ease with using a console 10 feet away from the patient to perform delicate surgeries. Instead of observing a bladder that is the size of a baseball to a softball with a plum-sized prostate gland attached to it several feet below his face on an operating table, the surgeon can now see in high-definition 3D a bladder that is the size of a soccer ball with a prostate gland the size of a baseball directly in front of his face.
Pre-Operation Procedures
After my initial check-in at 5:30 in the morning, I was given a gown, prepped in the pre-op room, hooked up to several IVs, and wheeled into the operating room on a gurney. Quite frankly, I don’t remember anything after the pre-op. I could have been signing over my first-born and my house for all I knew (Figure 7) as everyone in the operating room was crowding around me.
FIGURE 7. Being wheeled into the operating room on a gurney.
Before the actual procedure, I was transferred to an operating table and my body was tipped head down to allow my gastrointestinal system to move away from the area of the operation. The anesthesiologist had to adjust the respiratory pressure because of this position to keep my lungs at full capacity. Figure 8 shows how I was draped in cloth and clear plastic in preparation for the surgery.
FIGURE 8. 'Out cold' on the table, draped in cloths, and ready for the robot.
How the EndoWrist Surgical Instruments Work
Before talking about the actual surgery, let me describe the instruments and how they work when attached to the robot. Figure 9 shows three of the EndoWrist instruments given to me by my surgeon.

FIGURE 9. Three EndoWrist instruments; one with the cover removed and one with the interconnecting disks shown.
At $2K to $2.5K each, these instruments can only be used 10 times. This limit was set (not because they get worn out since most EndoWrists can last much longer) to insure a very safe limit for a device that is used within a human body. At this point, the tiny memory chip on the green circuit board inside the head of the EndoWrist (shown in Figure 10) has recorded each surgical use and it informs the da Vinci system that it can no longer be used (or it can be given to an overly-curious patient like me). The blue plastic port and attached clear tubing above the circuit board allows a saline solution to be circulated at the EndoWrist’s working site.

FIGURE 10. EndoWrist head showing cable reels.
Before I was given the EndoWrist instruments, I assumed that the actual end-effector received its mechanical movement via a series of rotating carbon fiber tubes; the smallest fitting within a slightly larger diameter tube, and so on. I discovered that the EndoWrist uses four small reels that move four stainless steel cables in or out that run up the carbon fiber tube to the end-effector. The largest diameter reel (# 1 in Figure 10) moves a short cable that runs to another smaller reel about 5 mm below, whose axis is 90° to the larger and faces down the outer tube; this causes the whole tube and end-effector to rotate almost 520°. The cables from two smaller reels at the far end of the blue attachment box (#2 and #3 in Figure 10) are each fastened to one of the two sides of forceps or scissors. Rather than have one reel open and close both halves of a pair of forceps or scissor tips, one reel/cable controls each half of the ‘jaws’ and allows the forceps or scissors to swivel and cut/pinch in a 200°+ arc. The fourth small reel’s (#4 in Figure 10) set of cables moves the tip in a 180º arc at a 90° angle to the jaw motion.
So, the surgeon has four degrees of freedom with the four cable reels, and a fifth axis which gives the robot the ability to move the instrument in and out along the axis of the tube. There are actually two more axes of motion — though limited — with the ability to tilt the instrument back and forth in an x-y motion. With a choice of three instruments to manipulate, hold, cut, pinch, and perform other functions, plus a high definition, magnified, and stereoscopic view of the operating site through the EndoScope camera system, many surgeons say that “this is the only way to perform delicate surgeries such as a prostatectomy or hysterectomy.” Another plus over handheld instruments using arthroscopic or laparoscopic procedures is that the surgeon does not have to hold his or her various instruments in a certain position for long periods of time — the robot does it. Doctors can rest a moment and determine the next step in the operation.
Positioning of the Robotic EndoWrist End-effectors
You may be wondering how the instruments are positioned into the surgery area. Obviously, a surgeon cannot have the robot blindly insert an instrument into a patient. The instruments must be positioned at the operation site without damaging nearby tissue or organs. Trocars are the guiding tubes that the surgeon inserts through the abdominal wall into the operating area. Figure 11 shows a typical trocar with the pointed insert.

FIGURE 11. A typical trocar 'tube' to allow instruments to easily enter the body. The inner pointed part is removed after insertion.
Figure 12 shows my personal trocar sites; these are the small entry sites for the surgical instruments. Six small incisions were made in my skin. The photo looks a bit scary but the scars have all but disappeared. This is a delicate manual procedure because the surgeon must not puncture any arteries, veins, or organs. The pointed end of the trocar moves slowly through tissue to the operation site; the point is then collapsed and removed so the surgeon can later insert, manipulate, and remove the EndoWrist instrument.
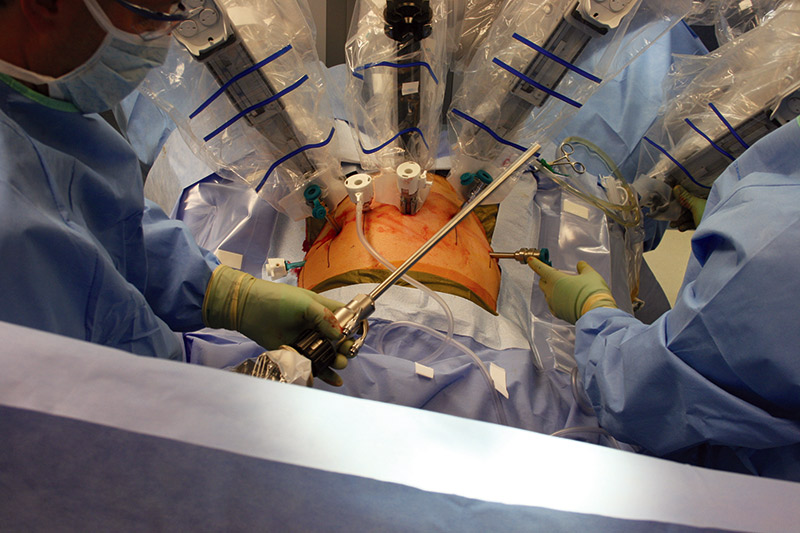
FIGURE 12. The robot is aligned with the trocars.
My surgeon used trocars of several different sizes to create entry sites ranging from 5, 8, and 12 mm in diameter. The largest incision (12 mm which was later extended to 4-5 cm) that was made on my body was just above my navel; this was first used for the camera, and then was extended to remove my prostate and seminal vesicles.
Smaller trocars were used for the 8 mm EndoWrist instruments, then another was used as a drain. A second 12 mm trocar was used as an assistant port.
Notice how my abdomen is slightly inflated with CO2 gas through the attached clear tubing to allow an open view of the prostate site and operating area with the endoscope TV camera. The first trocar in was the ‘optical’ trocar so my surgeon could see what was happening with the insertion of the remaining trocars and the required operating instruments.
The Surgery Procedures
With the trocars in place, a catheter hooked up, the abdomen inflated at 15 mm/Hg of pressure, and the endoscope video and lighting in place, the da Vinci robot is brought over and locked into position over the patient. Each small step is certainly more complex than the following descriptions, but I’m just giving a quick timeline. The required EndoWrist instruments for the first part of the surgery are slowly inserted through the trocar and into the operation area. The surgeon first severs the vas deferens and the seminal vesicles, and removes them. The bladder is lowered to reach the prostate. The delicate procedure of severing the prostate from the bladder is performed as the surgeon must not damage the bladder’s sphincter muscle that acts as a valve for empting the bladder. The urethra is carefully severed from the other side of the prostate and the gland is put into a small ‘zip lock bag’ to be removed. The urethra is then stitched to the sphincter muscle to get the patient’s plumbing back in order.
All of this takes from two and a half to three hours. Just imagine the many hundreds of critical movements that the surgeon has to perform! There are many instruments that he/she must use, such as needle drivers, forceps, scissors, and many others. Figure 13 shows a close-up view of my three EndoWrist instrument’s ‘working’ ends.

FIGURE 13. Three different EndoWrist end-effectors.
Figure 14 shows a close-up view of a needle driver EndoWrist. Figure 15 shows a small assortment of the different types of EndoWrist instruments available.

FIGURE 14. EndoWrist needle driver.

FIGURE 15. A small array of instruments.
Notice how the cables are attached to the different end instruments. Each new instrument is attached to the robot’s arm and inserted into the trocar which has a seal at the bottom of the tube. Figure 16 shows a basic suturing procedure under magnification.
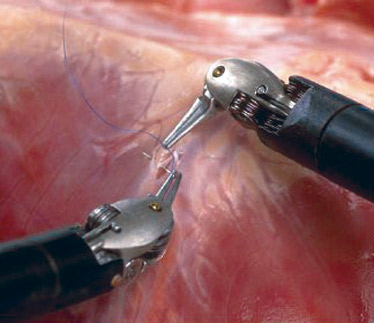
FIGURE 16. A basic suture procedure.
My operation could have been accomplished by any of the methods I talked about earlier. I preferred the da Vinci robot-assisted surgery, mainly for the ability of my surgeon to do the delicate procedures such as nerve sparing and small suturing to allow me a quick recovery process. This minimally invasive procedure allowed me to be up and walking the afternoon of my surgery, and going home the next day. Yes, there were a few more days of rest and simple exercising, but I was soon doing daily hikes with my wife.
Final Thoughts
In a demonstration, engineering and post-grad computer science students at Johns Hopkins University in Baltimore modified a Da Vinci surgical system console and used it as a workstation to control an industrial robot at NASA Goddard Flight Center 30 miles away. The same high definition video link used for surgery allowed operators to see the interior of a satellite, giving them the same virtual sensation of ‘being there’ that surgeons experience. It is hoped that the repair, refueling, and maintenance of on-orbit satellites may not require the presence of astronauts in the near future. NASA has the Robonaut 2, and possibly it can be used to hand the da Vinci robot tools as needed for space-borne repairs.
I am indebted to my surgeon, Dr. Michael Lipke (a urologist with Kaiser Permanente), who endured my incessant medical and technical questions, and did a great surgery for me, as well as the many nurses, anesthesiologist, assistant surgeon, and other caregivers at Providence St. Vincent Medical Center in Portland. I would also like to thank Providence St. Vincent for allowing more than 60 photographs to be taken during my operation. I appreciate all of the help that Intuitive Surgical gave to me, and steering me to the right people. This was a most unique and educational experience for me. SV
Article Comments